Roles of bioactive sphingolipids in cancer biology and therapeutics
- PMID: 18751921
- PMCID: PMC2636716
- DOI: 10.1007/978-1-4020-8831-5_16
Roles of bioactive sphingolipids in cancer biology and therapeutics
Abstract
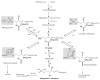
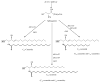
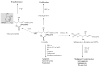
In this chapter, roles of bioactive sphingolipids in the regulation of cancer pathogenesis and therapy will be reviewed. Sphingolipids have emerged as bioeffector molecules, which control various aspects of cell growth, proliferation, and anti-cancer therapeutics. Ceramide, the central molecule of sphingolipid metabolism, generally mediates anti-proliferative responses such as inhibition of cell growth, induction of apoptosis, and/or modulation of senescence. On the other hand, sphingosine 1-phosphate (S1P) plays opposing roles, and induces transformation, cancer cell growth, or angiogenesis. A network of metabolic enzymes regulates the generation of ceramide and S1P, and these enzymes serve as transducers of sphingolipid-mediated responses that are coupled to various exogenous or endogenous cellular signals. Consistent with their key roles in the regulation of cancer growth and therapy, attenuation of ceramide generation and/or increased S1P levels are implicated in the development of resistance to drug-induced apoptosis, and escape from cell death. These data strongly suggest that advances in the molecular and biochemical understanding of sphingolipid metabolism and function will lead to the development of novel therapeutic strategies against human cancers, which may also help overcome drug resistance.
Figures
Similar articles
-
Sphingolipids and cancer: ceramide and sphingosine-1-phosphate in the regulation of cell death and drug resistance.Future Oncol. 2010 Oct;6(10):1603-24. doi: 10.2217/fon.10.116. Future Oncol. 2010. PMID: 21062159 Free PMC article. Review.
-
Sphingosine kinase/sphingosine 1-phosphate signaling in cancer therapeutics and drug resistance.Handb Exp Pharmacol. 2013;(216):3-27. doi: 10.1007/978-3-7091-1511-4_1. Handb Exp Pharmacol. 2013. PMID: 23563649 Review.
-
The role of sphingosine-1-phosphate and ceramide-1-phosphate in calcium homeostasis.Curr Opin Investig Drugs. 2008 Nov;9(11):1192-205. Curr Opin Investig Drugs. 2008. PMID: 18951299 Review.
-
"Inside-out" signaling of sphingosine-1-phosphate: therapeutic _targets.Pharmacol Rev. 2008 Jun;60(2):181-95. doi: 10.1124/pr.107.07113. Epub 2008 Jun 13. Pharmacol Rev. 2008. PMID: 18552276 Free PMC article. Review.
-
Sphingolipid metabolism in cancer signalling and therapy.Nat Rev Cancer. 2018 Jan;18(1):33-50. doi: 10.1038/nrc.2017.96. Epub 2017 Nov 17. Nat Rev Cancer. 2018. PMID: 29147025 Free PMC article. Review.
Cited by
-
Utilization of metabolomics to identify serum biomarkers for hepatocellular carcinoma in patients with liver cirrhosis.Anal Chim Acta. 2012 Sep 19;743:90-100. doi: 10.1016/j.aca.2012.07.013. Epub 2012 Jul 20. Anal Chim Acta. 2012. PMID: 22882828 Free PMC article.
-
Global Changes in Lipid Profiles of Mouse Cortex, Hippocampus, and Hypothalamus Upon p53 Knockout.Sci Rep. 2016 Nov 7;6:36510. doi: 10.1038/srep36510. Sci Rep. 2016. PMID: 27819311 Free PMC article.
-
Cancer Prevention and Therapy with Polyphenols: Sphingolipid-Mediated Mechanisms.Nutrients. 2018 Jul 21;10(7):940. doi: 10.3390/nu10070940. Nutrients. 2018. PMID: 30037082 Free PMC article. Review.
-
A hypothesis concerning a potential involvement of ceramide in apoptosis and acantholysis induced by pemphigus autoantibodies.Dermatol Res Pract. 2010;2010:702409. doi: 10.1155/2010/702409. Epub 2010 May 18. Dermatol Res Pract. 2010. PMID: 20585604 Free PMC article.
-
Sphingolipids and Lymphomas: A Double-Edged Sword.Cancers (Basel). 2022 Apr 19;14(9):2051. doi: 10.3390/cancers14092051. Cancers (Basel). 2022. PMID: 35565181 Free PMC article. Review.
References
-
- Abe A, Radin NS, Shayman JA, Wotring LL, Zipkin RE, Sivakumar R, Ruggieri JM, Carson KG, Ganem B. Structural and stereochemical studies of potent inhibitors of glucosylceramide synthase and tumor cell growth. J Lipid Res. 1995;36:611–621. - PubMed
-
- Akao Y, Banno Y, Nakagawa Y, Hasegawa N, Kim TJ, Murate T, Igarashi Y, Nozawa Y. High expression of sphingosine kinase 1 and S1P receptors in chemotherapy-resistant prostate cancer PC3 cells and their camptothecin-induced up-regulation. Biochem Biophys Res Commun. 2006;342:1284–1290. - PubMed
-
- Alexander S, Min J, Alexander H. Dictyostelium discoideum to human cells: pharmacogenetic studies demonstrate a role for sphingolipids in chemoresistance. Biochim Biophys Acta. 2006;1760:301–309. - PubMed
-
- Andrieu-Abadie N, Levade T. Sphingomyelin hydrolysis during apoptosis. Biochim Biophys Acta. 2002;1585:126–134. - PubMed
-
- Argraves KM, Wilkerson BA, Argraves WS, Fleming PA, Obeid LM, Drake CJ. Sphingosine-1-phosphate signaling promotes critical migratory events in vasculogenesis. J Biol Chem. 2004;279:50580–50590. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
