Overcoming endocrine resistance due to reduced PTEN levels in estrogen receptor-positive breast cancer by co-_targeting mammalian _target of rapamycin, protein kinase B, or mitogen-activated protein kinase kinase
- PMID: 25212826
- PMCID: PMC4303114
- DOI: 10.1186/s13058-014-0430-x
Overcoming endocrine resistance due to reduced PTEN levels in estrogen receptor-positive breast cancer by co-_targeting mammalian _target of rapamycin, protein kinase B, or mitogen-activated protein kinase kinase
Abstract
Introduction: Activation of the phosphatidylinositol 3-kinase (PI3K) pathway in estrogen receptor α (ER)-positive breast cancer is associated with reduced ER expression and activity, luminal B subtype, and poor outcome. Phosphatase and tensin homolog (PTEN), a negative regulator of this pathway, is typically lost in ER-negative breast cancer. We set out to clarify the role of reduced PTEN levels in endocrine resistance, and to explore the combination of newly developed PI3K downstream kinase inhibitors to overcome this resistance.
Methods: Altered cellular signaling, gene expression, and endocrine sensitivity were determined in inducible PTEN-knockdown ER-positive/human epidermal growth factor receptor 2 (HER2)-negative breast cancer cell and/or xenograft models. Single or two-agent combinations of kinase inhibitors were examined to improve endocrine therapy.
Results: Moderate PTEN reduction was sufficient to enhance PI3K signaling, generate a gene signature associated with the luminal B subtype of breast cancer, and cause endocrine resistance in vitro and in vivo. The mammalian _target of rapamycin (mTOR), protein kinase B (AKT), or mitogen-activated protein kinase kinase (MEK) inhibitors, alone or in combination, improved endocrine therapy, but the efficacy varied by PTEN levels, type of endocrine therapy, and the specific inhibitor(s). A single-agent AKT inhibitor combined with fulvestrant conferred superior efficacy in overcoming resistance, inducing apoptosis and tumor regression.
Conclusions: Moderate reduction in PTEN, without complete loss, can activate the PI3K pathway to cause endocrine resistance in ER-positive breast cancer, which can be overcome by combining endocrine therapy with inhibitors of the PI3K pathway. Our data suggests that the ER degrader fulvestrant, to block both ligand-dependent and -independent ER signaling, combined with an AKT inhibitor is an effective strategy to test in patients.
Figures
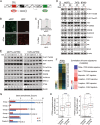
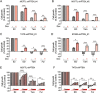
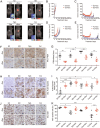


Comment in
-
_targeting PTEN-defined breast cancers with a one-two punch.Breast Cancer Res. 2015 Apr 8;17(1):51. doi: 10.1186/s13058-015-0566-3. Breast Cancer Res. 2015. PMID: 25888162 Free PMC article.
Similar articles
-
Preclinical modeling of combined phosphatidylinositol-3-kinase inhibition with endocrine therapy for estrogen receptor-positive breast cancer.Breast Cancer Res. 2011 Mar 1;13(2):R21. doi: 10.1186/bcr2833. Breast Cancer Res. 2011. PMID: 21362200 Free PMC article.
-
Impact of dual mTORC1/2 mTOR kinase inhibitor AZD8055 on acquired endocrine resistance in breast cancer in vitro.Breast Cancer Res. 2014 Jan 23;16(1):R12. doi: 10.1186/bcr3604. Breast Cancer Res. 2014. PMID: 24457069 Free PMC article.
-
AKT Antagonist AZD5363 Influences Estrogen Receptor Function in Endocrine-Resistant Breast Cancer and Synergizes with Fulvestrant (ICI182780) In Vivo.Mol Cancer Ther. 2015 Sep;14(9):2035-48. doi: 10.1158/1535-7163.MCT-15-0143. Epub 2015 Jun 26. Mol Cancer Ther. 2015. PMID: 26116361
-
Mechanism of resistance to endocrine therapy in breast cancer: the important role of PI3K/Akt/mTOR in estrogen receptor-positive, HER2-negative breast cancer.Breast Cancer. 2018 Jul;25(4):392-401. doi: 10.1007/s12282-017-0812-x. Epub 2017 Oct 31. Breast Cancer. 2018. PMID: 29086897 Review.
-
_targeting the PI3K/AKT/mTOR pathway in estrogen receptor-positive breast cancer.Cancer Treat Rev. 2014 Aug;40(7):862-71. doi: 10.1016/j.ctrv.2014.03.004. Epub 2014 Mar 26. Cancer Treat Rev. 2014. PMID: 24774538 Review.
Cited by
-
Pictilisib for oestrogen receptor-positive, aromatase inhibitor-resistant, advanced or metastatic breast cancer (FERGI): a randomised, double-blind, placebo-controlled, phase 2 trial.Lancet Oncol. 2016 Jun;17(6):811-821. doi: 10.1016/S1470-2045(16)00106-6. Epub 2016 May 4. Lancet Oncol. 2016. PMID: 27155741 Free PMC article. Clinical Trial.
-
Current Molecular Combination Therapies Used for the Treatment of Breast Cancer.Int J Mol Sci. 2022 Sep 20;23(19):11046. doi: 10.3390/ijms231911046. Int J Mol Sci. 2022. PMID: 36232349 Free PMC article. Review.
-
Deubiquitinases and the new therapeutic opportunities offered to cancer.Endocr Relat Cancer. 2015 Feb;22(1):T35-54. doi: 10.1530/ERC-14-0516. Endocr Relat Cancer. 2015. PMID: 25605410 Free PMC article. Review.
-
The oral selective oestrogen receptor degrader (SERD) AZD9496 is comparable to fulvestrant in antagonising ER and circumventing endocrine resistance.Br J Cancer. 2019 Feb;120(3):331-339. doi: 10.1038/s41416-018-0354-9. Epub 2018 Dec 17. Br J Cancer. 2019. PMID: 30555156 Free PMC article.
-
Low PTEN levels and PIK3CA mutations predict resistance to neoadjuvant lapatinib and trastuzumab without chemotherapy in patients with HER2 over-expressing breast cancer.Breast Cancer Res Treat. 2018 Feb;167(3):731-740. doi: 10.1007/s10549-017-4533-9. Epub 2017 Nov 7. Breast Cancer Res Treat. 2018. PMID: 29110152 Free PMC article. Clinical Trial.
References
-
- Rimawi MF, Wiechmann LS, Wang YC, Huang C, Migliaccio I, Wu MF, Gutierrez C, Hilsenbeck SG, Arpino G, Massarweh S, Ward R, Soliz R, Osborne CK, Schiff R. Reduced dose and intermittent treatment with lapatinib and trastuzumab for potent blockade of the HER pathway in HER2/neu-overexpressing breast tumor xenografts. Clin Cancer Res. 2011;17:1351–1361. doi: 10.1158/1078-0432.CCR-10-1905. - DOI - PMC - PubMed
-
- Creighton CJ, Fu X, Hennessy BT, Casa AJ, Zhang Y, Gonzalez-Angulo AM, Lluch A, Gray JW, Brown PH, Hilsenbeck SG, Osborne CK, Mills GB, Lee AV, Schiff R: Proteomic and transcriptomic profiling reveals a link between the PI3K pathway and lower estrogen-receptor (ER) levels and activity in ER + breast cancer.Breast Cancer Res 2010, 12:R40., - PMC - PubMed
-
- Loi S, Haibe-Kains B, Majjaj S, Lallemand F, Durbecq V, Larsimont D, Gonzalez-Angulo AM, Pusztai L, Symmans WF, Bardelli A, Ellis P, Tutt AN, Gillett CE, Hennessy BT, Mills GB, Phillips WA, Piccart MJ, Speed TP, McArthur GA, Sotiriou C. PIK3CA mutations associated with gene signature of low mTORC1 signaling and better outcomes in estrogen receptor-positive breast cancer. Proc Natl Acad Sci U S A. 2010;107:10208–10213. doi: 10.1073/pnas.0907011107. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- R01 CA94118/CA/NCI NIH HHS/United States
- AI036211/AI/NIAID NIH HHS/United States
- S10RR024574/RR/NCRR NIH HHS/United States
- P30CA125123/CA/NCI NIH HHS/United States
- R01 CA151962/CA/NCI NIH HHS/United States
- P30 AI036211/AI/NIAID NIH HHS/United States
- P50 CA058183/CA/NCI NIH HHS/United States
- S10 RR024574/RR/NCRR NIH HHS/United States
- R01 CA094118/CA/NCI NIH HHS/United States
- P50 CA58183/CA/NCI NIH HHS/United States
- P30 CA125123/CA/NCI NIH HHS/United States
- P50 CA58207/CA/NCI NIH HHS/United States
- P50 CA058207/CA/NCI NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

