LATS2 Positively Regulates Polycomb Repressive Complex 2
- PMID: 27434182
- PMCID: PMC4951031
- DOI: 10.1371/journal.pone.0158562
LATS2 Positively Regulates Polycomb Repressive Complex 2
Abstract
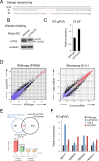
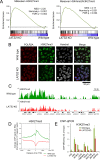
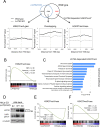
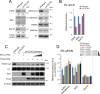
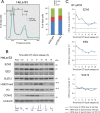
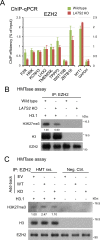
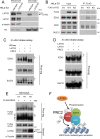
LATS2, a pivotal Ser/Thr kinase of the Hippo pathway, plays important roles in many biological processes. LATS2 also function in Hippo-independent pathway, including mitosis, DNA damage response and epithelial to mesenchymal transition. However, the physiological relevance and molecular basis of these LATS2 functions remain obscure. To understand novel functions of LATS2, we constructed a LATS2 knockout HeLa-S3 cell line using TAL-effector nuclease (TALEN). Integrated omics profiling of this cell line revealed that LATS2 knockout caused genome-wide downregulation of Polycomb repressive complex 2 (PRC2) and H3K27me3. Cell-cycle analysis revealed that downregulation of PRC2 was not due to cell cycle aberrations caused by LATS2 knockout. Not LATS1, a homolog of LATS2, but LATS2 bound PRC2 on chromatin and phosphorylated it. LATS2 positively regulates histone methyltransferase activity of PRC2 and their expression at both the mRNA and protein levels. Our findings reveal a novel signal upstream of PRC2, and provide insight into the crucial role of LATS2 in coordinating the epigenome through regulation of PRC2.
Conflict of interest statement
Figures
Similar articles
-
Polycomb repressive complex 2 epigenomic signature defines age-associated hypermethylation and gene expression changes.Epigenetics. 2015;10(6):484-95. doi: 10.1080/15592294.2015.1040619. Epub 2015 Apr 16. Epigenetics. 2015. PMID: 25880792 Free PMC article.
-
miR-135b, upregulated in breast cancer, promotes cell growth and disrupts the cell cycle by regulating LATS2.Int J Oncol. 2016 May;48(5):1997-2006. doi: 10.3892/ijo.2016.3405. Epub 2016 Feb 22. Int J Oncol. 2016. PMID: 26934863
-
Deubiquitylase USP9X suppresses tumorigenesis by stabilizing large tumor suppressor kinase 2 (LATS2) in the Hippo pathway.J Biol Chem. 2018 Jan 26;293(4):1178-1191. doi: 10.1074/jbc.RA117.000392. Epub 2017 Nov 28. J Biol Chem. 2018. PMID: 29183995 Free PMC article.
-
The LATS1 and LATS2 tumor suppressors: beyond the Hippo pathway.Cell Death Differ. 2017 Sep;24(9):1488-1501. doi: 10.1038/cdd.2017.99. Epub 2017 Jun 23. Cell Death Differ. 2017. PMID: 28644436 Free PMC article. Review.
-
Recruiting polycomb to chromatin.Int J Biochem Cell Biol. 2015 Oct;67:177-87. doi: 10.1016/j.biocel.2015.05.006. Epub 2015 May 14. Int J Biochem Cell Biol. 2015. PMID: 25982201 Free PMC article. Review.
Cited by
-
Genome-wide interaction study of a proxy for stress-sensitivity and its prediction of major depressive disorder.PLoS One. 2018 Dec 20;13(12):e0209160. doi: 10.1371/journal.pone.0209160. eCollection 2018. PLoS One. 2018. PMID: 30571770 Free PMC article.
-
A feedback loop between plakophilin 4 and YAP signaling regulates keratinocyte differentiation.iScience. 2024 Aug 19;27(9):110762. doi: 10.1016/j.isci.2024.110762. eCollection 2024 Sep 20. iScience. 2024. PMID: 39286493 Free PMC article.
-
_targeted Disruption of Lats1 and Lats2 in Mice Impairs Testis Development and Alters Somatic Cell Fate.Int J Mol Sci. 2022 Nov 5;23(21):13585. doi: 10.3390/ijms232113585. Int J Mol Sci. 2022. PMID: 36362374 Free PMC article.
-
LATS1/2 kinases trigger self-renewal of cancer stem cells in aggressive oral cancer.Onco_target. 2019 Feb 1;10(10):1014-1030. doi: 10.18632/onco_target.26583. eCollection 2019 Feb 1. Onco_target. 2019. PMID: 30800215 Free PMC article.
-
Long noncoding RNA NEAT1 suppresses hepatocyte proliferation in fulminant hepatic failure through increased recruitment of EZH2 to the LATS2 promoter region and promotion of H3K27me3 methylation.Exp Mol Med. 2020 Mar;52(3):461-472. doi: 10.1038/s12276-020-0387-z. Epub 2020 Mar 10. Exp Mol Med. 2020. PMID: 32157157 Free PMC article.
References
-
- Visser S, Yang X. LATS tumor suppressor: a new governor of cellular homeostasis. Cell Cycle. 2010;9(19):3892–903. . - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

