Acetate Recapturing by Nuclear Acetyl-CoA Synthetase 2 Prevents Loss of Histone Acetylation during Oxygen and Serum Limitation
- PMID: 28099844
- PMCID: PMC5276806
- DOI: 10.1016/j.celrep.2016.12.055
Acetate Recapturing by Nuclear Acetyl-CoA Synthetase 2 Prevents Loss of Histone Acetylation during Oxygen and Serum Limitation
Abstract
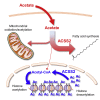
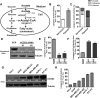
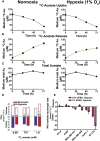
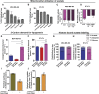
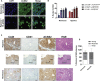
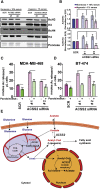
Acetyl-CoA is a key metabolic intermediate with an important role in transcriptional regulation. The nuclear-cytosolic acetyl-CoA synthetase 2 (ACSS2) was found to sustain the growth of hypoxic tumor cells. It generates acetyl-CoA from acetate, but exactly which pathways it supports is not fully understood. Here, quantitative analysis of acetate metabolism reveals that ACSS2 fulfills distinct functions depending on its cellular location. Exogenous acetate uptake is controlled by expression of both ACSS2 and the mitochondrial ACSS1, and ACSS2 supports lipogenesis. The mitochondrial and lipogenic demand for two-carbon acetyl units considerably exceeds the uptake of exogenous acetate, leaving it to only sparingly contribute to histone acetylation. Surprisingly, oxygen and serum limitation increase nuclear localization of ACSS2. We find that nuclear ACSS2 recaptures acetate released from histone deacetylation for recycling by histone acetyltransferases. Our work provides evidence for limited equilibration between nuclear and cytosolic acetyl-CoA and demonstrates that ACSS2 retains acetate to maintain histone acetylation.
Keywords: acetate; acetyl-CoA synthetase 2; cancer metabolism; enzyme localization; histone acetylation; histone deacetylation; hypoxia; lipogenesis; metabolite compartmentalization; stable isotope tracing.
Copyright © 2017 The Author(s). Published by Elsevier Inc. All rights reserved.
Figures

Similar articles
-
Nucleus-Translocated ACSS2 Promotes Gene Transcription for Lysosomal Biogenesis and Autophagy.Mol Cell. 2017 Jun 1;66(5):684-697.e9. doi: 10.1016/j.molcel.2017.04.026. Epub 2017 May 25. Mol Cell. 2017. PMID: 28552616 Free PMC article.
-
Acetyl-CoA synthases are essential for maintaining histone acetylation under metabolic stress during zygotic genome activation in pigs.J Cell Physiol. 2021 Oct;236(10):6948-6962. doi: 10.1002/jcp.30355. Epub 2021 Mar 8. J Cell Physiol. 2021. PMID: 33682931
-
Local histone acetylation by ACSS2 promotes gene transcription for lysosomal biogenesis and autophagy.Autophagy. 2017 Oct 3;13(10):1790-1791. doi: 10.1080/15548627.2017.1349581. Epub 2017 Aug 18. Autophagy. 2017. PMID: 28820290 Free PMC article.
-
Acetyl-CoA synthetase 2(ACSS2): a review with a focus on metabolism and tumor development.Discov Oncol. 2022 Jul 7;13(1):58. doi: 10.1007/s12672-022-00521-1. Discov Oncol. 2022. PMID: 35798917 Free PMC article. Review.
-
Acetate Revisited: A Key Biomolecule at the Nexus of Metabolism, Epigenetics and Oncogenesis-Part 1: Acetyl-CoA, Acetogenesis and Acyl-CoA Short-Chain Synthetases.Front Physiol. 2020 Nov 12;11:580167. doi: 10.3389/fphys.2020.580167. eCollection 2020. Front Physiol. 2020. PMID: 33281616 Free PMC article. Review.
Cited by
-
The interaction between the gut microbiota and dietary carbohydrates in nonalcoholic fatty liver disease.Exp Mol Med. 2021 May;53(5):809-822. doi: 10.1038/s12276-021-00614-x. Epub 2021 May 20. Exp Mol Med. 2021. PMID: 34017059 Free PMC article. Review.
-
Tailoring a CRISPR/Cas-based Epigenome Editor for Programmable Chromatin Acylation and Decreased Cytotoxicity.bioRxiv [Preprint]. 2024 Sep 22:2024.09.22.611000. doi: 10.1101/2024.09.22.611000. bioRxiv. 2024. PMID: 39345554 Free PMC article. Preprint.
-
Acetyl-CoA metabolism in cancer.Nat Rev Cancer. 2023 Mar;23(3):156-172. doi: 10.1038/s41568-022-00543-5. Epub 2023 Jan 19. Nat Rev Cancer. 2023. PMID: 36658431 Free PMC article. Review.
-
Acetate Production from Glucose and Coupling to Mitochondrial Metabolism in Mammals.Cell. 2018 Oct 4;175(2):502-513.e13. doi: 10.1016/j.cell.2018.08.040. Epub 2018 Sep 20. Cell. 2018. PMID: 30245009 Free PMC article.
-
Combining metabolic phenotype determination with metabolomics and transcriptional analyses to reveal pathways regulated by hydroxycarboxylic acid receptor 2.Discov Oncol. 2022 Jun 13;13(1):47. doi: 10.1007/s12672-022-00503-3. Discov Oncol. 2022. PMID: 35697980 Free PMC article.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

