Streptococcus mutans suppresses filamentous growth of Candida albicans through secreting mutanocyclin, an unacylated tetramic acid
- PMID: 35311622
- PMCID: PMC8942415
- DOI: 10.1080/21505594.2022.2046952
Streptococcus mutans suppresses filamentous growth of Candida albicans through secreting mutanocyclin, an unacylated tetramic acid
Abstract
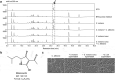
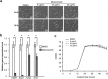
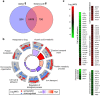
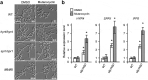
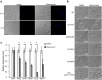
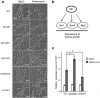
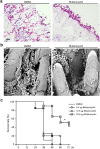
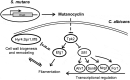
Fungi and bacteria often co-exist and physically or chemically interact with each other in their natural niches. This inter-kingdom species interaction is exemplified by the gram-positive bacterial pathogen Streptococcus mutans and opportunistic fungal pathogen Candida albicans, which co-exist in the human mouth. It has been demonstrated that the dynamic interaction between these two species plays a critical role in their virulence and biofilm development. In this study, we discovered that S. mutans represses filamentous development and virulence in C. albicans through secreting a secondary metabolite, mutanocyclin (a tetramic acid). Mutanocyclin functions by regulating the PKA catabolic subunit Tpk2 and its preferential binding _target Sfl1. Inactivation of Tpk2 in C. albicans results in an increased sensitivity to mutanocyclin, whereas overexpression of Tpk2 leads to an increased resistance. Dysfunction of SFL1 and its downstream _target genes overrides the hyphal growth defect caused by mutanocyclin. Further investigation demonstrates that three glycosylphosphatidylinositol (GPI)-anchored proteins (Spr1, Hyr4, and Iff8), associated with cell wall biogenesis and remodeling, and a set of filamentous regulators also contribute to the mutanocyclin response. We propose that both transcriptional regulation and cell wall composition contribute to mutanocyclin-mediated filamentous inhibition. This repressive effect of mutanocyclin could function as a natural regulator of filamentous development in C. albicans.
Keywords: Candida albicans; Streptococcus mutans; filamentous growth; mutanocyclin; transcriptional regulation.
Conflict of interest statement
No potential conflict of interest was reported by the author(s).
Figures

Similar articles
-
Tetramic Acids Mutanocyclin and Reutericyclin A, Produced by Streptococcus mutans Strain B04Sm5 Modulate the Ecology of an in vitro Oral Biofilm.Front Oral Health. 2022 Jan 7;2:796140. doi: 10.3389/froh.2021.796140. eCollection 2021. Front Oral Health. 2022. PMID: 35048077 Free PMC article.
-
mucG, mucH, and mucI Modulate Production of Mutanocyclin and Reutericyclins in Streptococcus mutans B04Sm5.J Bacteriol. 2022 May 17;204(5):e0004222. doi: 10.1128/jb.00042-22. Epub 2022 Apr 11. J Bacteriol. 2022. PMID: 35404110 Free PMC article.
-
Assessing the impact of curcumin on dual-species biofilms formed by Streptococcus mutans and Candida albicans.Microbiologyopen. 2019 Dec;8(12):e937. doi: 10.1002/mbo3.937. Epub 2019 Sep 27. Microbiologyopen. 2019. PMID: 31560838 Free PMC article.
-
Current and prospective therapeutic strategies: tackling Candida albicans and Streptococcus mutans cross-kingdom biofilm.Front Cell Infect Microbiol. 2023 May 11;13:1106231. doi: 10.3389/fcimb.2023.1106231. eCollection 2023. Front Cell Infect Microbiol. 2023. PMID: 37249973 Free PMC article. Review.
-
Suppression of hyphal formation and virulence of Candida albicans by natural and synthetic compounds.Biofouling. 2021 Jul;37(6):626-655. doi: 10.1080/08927014.2021.1948538. Epub 2021 Jul 20. Biofouling. 2021. PMID: 34284656 Review.
Cited by
-
[Latest Findings on Polyketides/Non-ribosomal Peptides That Are Secondary Metabolites of Streptococcus mutans].Sichuan Da Xue Xue Bao Yi Xue Ban. 2023 May;54(3):685-691. doi: 10.12182/20230560302. Sichuan Da Xue Xue Bao Yi Xue Ban. 2023. PMID: 37248606 Free PMC article. Review. Chinese.
-
Candida albicans CHK1 gene regulates its cross-kingdom interactions with Streptococcus mutans to promote caries.Appl Microbiol Biotechnol. 2022 Nov;106(21):7251-7263. doi: 10.1007/s00253-022-12211-7. Epub 2022 Oct 5. Appl Microbiol Biotechnol. 2022. PMID: 36195704
-
Human-associated bacteria adopt an unusual route for synthesizing 3-acetylated tetramates for environmental adaptation.Microbiome. 2023 May 5;11(1):97. doi: 10.1186/s40168-023-01548-4. Microbiome. 2023. PMID: 37147735 Free PMC article.
-
Surface adherence and vacuolar internalization of bacterial pathogens to the Candida spp. cells: Mechanism of persistence and propagation.J Adv Res. 2023 Nov;53:115-136. doi: 10.1016/j.jare.2022.12.013. Epub 2022 Dec 23. J Adv Res. 2023. PMID: 36572338 Free PMC article. Review.
-
Streptococcus mutans supernatant affects the virulence of Candida albicans.Braz J Microbiol. 2024 Mar;55(1):365-374. doi: 10.1007/s42770-023-01198-6. Epub 2023 Dec 2. Braz J Microbiol. 2024. PMID: 38040990 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
